Note that there is no clear division between metallic and non-metallic character. Scientist Date Robert 1661 Boyle Johann 1829 Döbereiner Contribution to the development of the. Fluorine gas reacts explosively with many other elements and compounds, and is considered to be one of the most dangerous known substances. The Basics: Periodic Table of Elements Part I: The History of the Periodic Table Visit Resource 1: Development of Elements and the Periodic Table to discover the contributions of some of the individuals that helped develop the periodic table: Approx. /caesium-cs--chemical-element--3d-rendering-isolated-on-black-background-922704282-5adb6de08e1b6e0037170151.jpg)
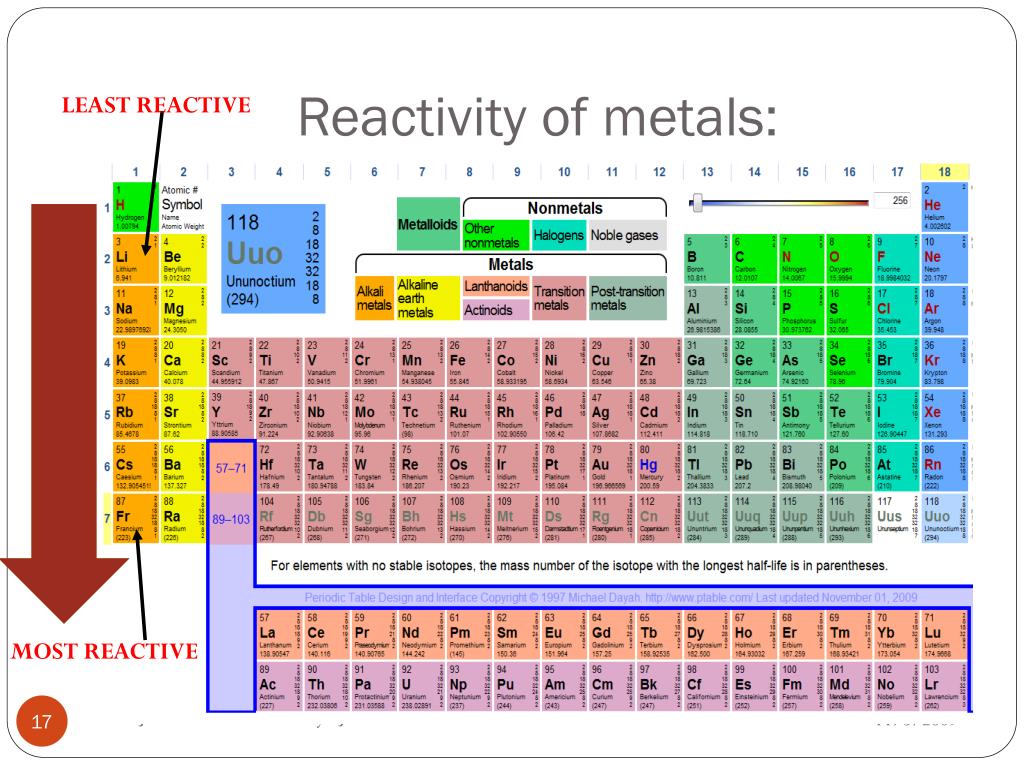
It is not found in nature as a free element. The two columns of elements in the periodic table that contain the most reactive elements are columns 1 and 17, the alkali metals and halogens respectively. 
Since the noble gases are a special group because of their lack of reactivity, the element fluorine is the most reactive nonmetal. The most reactive nonmetals reside in the upper right portion of the periodic table. 
Nonmetals tend to gain electrons in chemical reactions, and have a high attraction for electrons within a compound. In addition, the atomic radius increases going down a group, placing the outer electrons further away from the nucleus and making that electron less attracted by the nucleus. Since the ionization energy decreases going down a group (or increases going up a group), the increased ability for metals lower in a group to lose electrons makes them more reactive. The metallic character increases as you go down a group. It should not be confused with 'group VIIIA' in the CAS system, which is group 18 (current IUPAC), the noble gases. ' Group 8' is the modern standard designation for this group, adopted by the IUPAC in 1990. Francium is most reactive element in periodic table. Reactivity of metals is based on processes such as the formation of halide compounds with halogens, and how easily the element displaces hydrogen from dilute acids. Group 8 is a group (column) of chemical elements in the periodic table.It consists of iron (Fe), ruthenium (Ru), osmium (Os) and hassium (Hs). Fluorine is the most reactive element in the periodic table, forming compounds with every other element except helium, neon, and argon. (Credit: User:Mirek2/Wikimedia Commons Source: Commons Wikimedia, Periodic Trends(opens in new window) License: Public Domain) The activity series is a list of elements in decreasing order of their reactivity. Furthermore, we’ll deal with some of the more complex metallic alloys, such as silver and gold. Along with this knowledge, it is also extremely important to remember the chemical properties of Fluorine, a lot of which differ greatly from other halogen elements.\): Trends in behaviors of elements. In The Periodic Table Most Reactive Metals Are Found There are numerous teams of metals within the Periodic Table, and this short article will check out the principle categories of these elements. NOTE: It is extremely important to be familiar with the trends of electronegativity and size in the halogen group to be able to answer this question. The halogens also form compounds with carbon easily organic molecules containing carbon are often known as alkyl halides, or organohalides, and have many different household and industrial uses.Īn extremely chemically electronegative gas which is toxic in nature with atoms of the smallest size when compared to any other halogen, Fluorine is the most reactive element present in Group VIIA and the most reactive non-metal present in the periodic table. They are usually found in combination with various metals in minerals, or in combination with other non-metals in molecular compounds. The halogens are extremely reactive, and are not found naturally in their elemental forms. In combination with other non-metals, the halogens form compounds through covalent bonding. This is one electron away from having a full octet of eight electrons, so these elements tend to form anions having -1 charges, known as halides: fluoride, F- chloride, Cl-, bromide, Br-, and iodide, I. The Group 7A elements have seven valence electrons in their highest-energy orbitals ($ns^2np^5$). To help us answer this question, let us first go through the properties of halogens and then with the trend of chemical properties in the group, determine the most reactive of them all. Now that you have this information, try thinking of all the trends in chemical properties while moving from top to bottom in a group. Hint: Group VIIA is the group of the periodic table which contains halogens.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
